By Shelagh M. Szabo
Interest in the inclusion of patient perspectives in health research is growing across all aspects of the field, including health technology assessment (HTA). A recent international joint task group establishing a definition of HTA noted that among the varied dimensions of value considered when assessing health technologies, wider implications for the patient, relatives, caregivers, and the population are frequently now included.[1] This type of information is often gathered qualitatively, directly from patients and caregivers and efforts to formalize how this type of qualitative evidence can inform HTA are ongoing.
Understanding how much qualitative data are currently being included in HTA submissions and the quality of these data will be important as agencies seek to develop frameworks for expanding patient engagement with HTA. To that end, with colleagues in the School of Health and Wellbeing at the University of Glasgow, we recently characterized the contemporary use of qualitative data provided as patient-based evidence within HTA submissions to the National Institute for Health and Care Excellence (NICE) and Canadian Agency for Drugs and Technologies in Health (CADTH). This review, which has just been published in the International Journal of Technology Assessment in Healthcare, looked at the methods employed, quality of data generated, and the objectives and topics described.
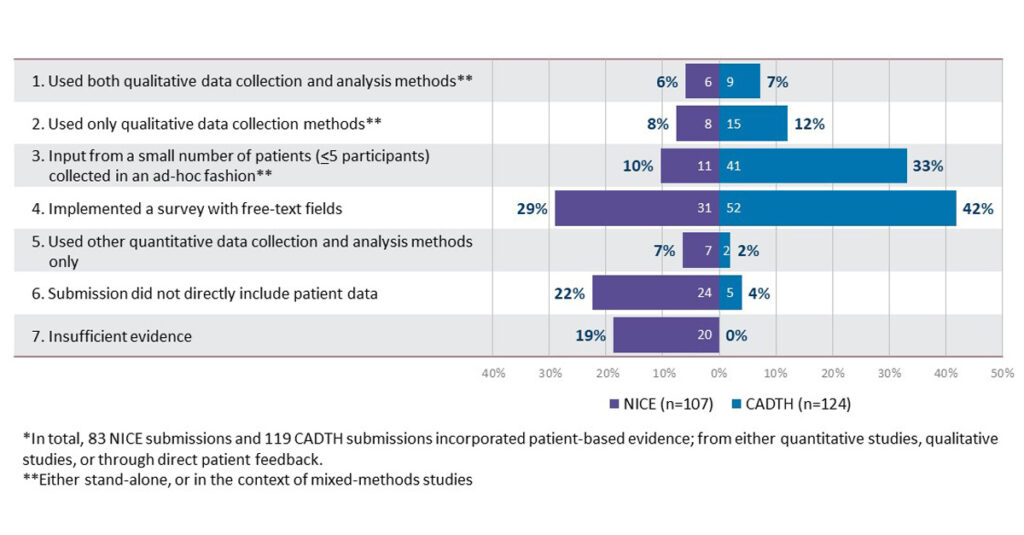
What we found was that while patient-based evidence was common in the submissions reviewed, only 13% of NICE and 19% of CADTH submissions reviewed involved formal qualitative data collection methods (generally one-to-one interviews or focus groups). Use of formal qualitative analysis methods was even rarer (thematic analysis was used exclusively) and reporting on the results of these analyses tended to be brief. Among these few from which it was possible to conduct a formal assessment using the CASP checklist, quality scores were generally moderate (see figure). These findings highlight the importance of direction from decision-makers about the types of qualitative evidence most useful to submit, and how these data should be synthesized and communicated to help inform decision making.
Our finding suggest if the increasing interest in providing patient-based evidence for HTA is to translate into impactful contributions, the focus needs to be on collecting and analyzing these data in a systematic and rigorous way to help ensure their usefulness and credibility. Guidance from decision-makers around key topics or areas for investigation will be helpful in the future for ensuring that that patient voices are clearly heard.
If you are interested in finding out more, I invite you to read our publication, “The extent and quality of qualitative evidence included in health technology assessments: a review of submissions to NICE and CADTH”
References
- O’Rourke B, Oortwijn W, Schuller T, International Joint Task G. The new definition of health technology assessment: A milestone in international collaboration. Int J Technol Assess Health Care. 2020;36(3):187-90.
